Hot Melt Extrusion in the Pharmaceutical Industry
Hot Melt Extrusion is increasingly being adopted in the Pharmaceutical industry as more and more drug ingredients, particularly Pharmaceutical APIs, are either poorly soluble or unstable during traditional processing using wet granulation.
By dispersing and mixing APIs with thermoplastic binders and/or polymers at the molecular level a solid solution (polymer matrix) is formed without the generation of dust, enabling drug delivery of poorly water-soluble or lipophilic APIs.

The Hot Melt Extrusion Process
During the hot melt extrusion (HME) process a powder blend of API and polymers are fed into a heated processing zone within the extruder. Twin screws impart a high shear and mixing action which disperses the API into a polymer matrix. The mix is forced through an opening to create an extrudate, which is then rapidly cooled so that it solidifies. If appropriate, the extrudate can be directly shaped into tablet form. However, it is more typical for extrudates to be further processed (i.e. milled) into a powder before it moves on down the solid dosage processing steps.
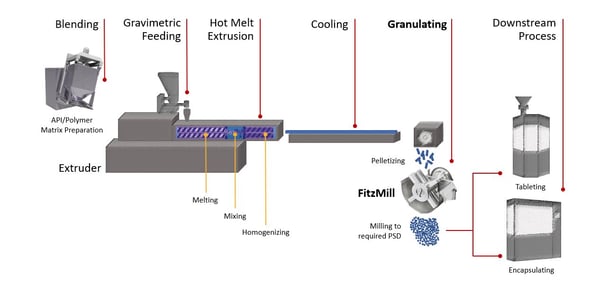
Compared to traditional pharmaceutical batch processing, this technology offers the advantages of product uniformity and higher manufacturing efficiency. With the incorporation of inline process analytical technology (PAT), individual parameters such as feed rate, screw speed, barrel temperature and vacuum can be adjusted independently to achieve the desired product quality targets.
Advantages of Hot Melt Extrusion
Manufacturers have found there are some significant advantages of adopting the HME process:
- It is an economical and efficient process that reduces production time as there is shorter material residence time
- The process is scalable with tighter process control
- The equipment has a smaller footprint compared to the traditional systems so reduces the burden on clean room needs
- It can be used to overcome solubility limitations, which will improve bioavailability by creating controlled release extrudates.
How to Sensitively Mill Hot Melt Extrusion
Since melt granulation is a size-enlargement method, the output material is not often within the ideal particle size range for direct introduction into compression machinery such as tablet presses. Therefore, it requires further processing.
When milling the extrudate, it is important to create a powder that has good flow properties and the desired bulk density needed to create a good-quality tablet. Thus, the choice of equipment is vital as it will significantly impact the quality of the final product.
But here lies a predicament. The low-temperature polymers and waxes used to preserve the efficacy of the active pharmaceutical ingredient are prone to re-melting or softening in the size reduction process if too much heat is produced in the milling effort, this can lead to an unwelcome extrusion effect.
A comment we often hear from companies, that ask for our help, is that they had started milling with simple technology, which is durable enough to handle the tough polymer, but they have found that it takes days to achieve results. This just isn’t practical as a manufacturing process step.
We have been able to bring all our milling expertise forth to help solve this issue. We are fortunate that both Fitzpatrick and Quadro are part of the same corporation, so we can determine the ideal mill for the circumstances.
Hot Melt Extrusion Equipment
The FitzMillTM D6Ax Hammer Mill is an ideal solution because the process material is introduced to a rotating blade. Although the blade rotates within a chamber, this chamber is designed to be away from the tip of the blades creating a relief space (hence the term in-air milling has been coined).
To mitigate the heat of impact, Fitzmills can come equipped with chilled water jackets which are applied in strategic locations. Since most of the size reduction is performed by the impact of the process material and the blades, the sizing screen can be much larger than the desired particle top size. The large aperture screen precludes blinding and the aforementioned extrusion effect.
The result is a robust milling process that can be used on heat-sensitive melt-extruded or melt- agglomerated material.
The Quadro Comil® High Energy H20 performs well when the pelletized material or ‘lentils’ (as these extrusions are sometimes referred to) are easier to mill. That is when the extrusions do not require the same in-air impact that is delivered by a FitzMill. Rather they can be sized reduced through attrition between a high-speed rotating impeller and a conical screen. With the H20 Comil, the use of cryo conditions is sometimes necessary to lower the glass transition temperature of the polymer and prevent screen blinding.
Help With Hot Melt Extrusion Technology
If you would like help choosing the right mill to complement your Hot Melt Extrusion technique, then get in touch with our experts who will be able to give you clear advice.



